Product Specifications
General Specifications
Description: Designed to be secure to the proximal portion of a guide wire with a
diameter of 0.009”, 0.014” or 0.018”. This device should only be used by
physicians trained in invasive vascular procedures.
Intended use: Vascular procedures in conjunction with interventional and/or
diagnostic devices to facilitate the steering of the guidewire within vascular
anatomy.
Single Use: Not designed to be re-sterilized or reused on another patient.
Sterilization Method: Electron Beam Irradiation, Non-Pyrogenic.
Bore Size: 0.009-0.022”*
Latex Free: Yes

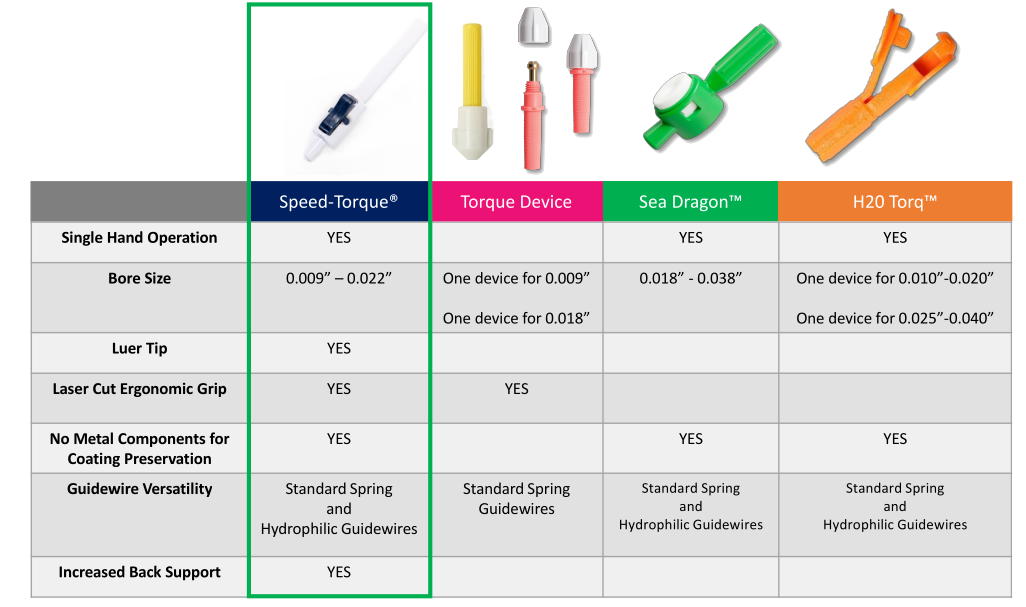
Speed-Torque Product Comparison
Regulatory Summary
Establishment Registration & Device Listing
Proprietary Name: Speed-Torque®
Classification Name: WIRE, GUIDE, CATHETER, EXEMPT
FDA Product Code: PTL
FDA Classification: Class II, 510(k) Exempt
Regulation Medical Specialty: Cardiovascular
Regulation Number: 870.1330
GMP Exempt: No
Clinically Differentiated
The Speed-Torque provides numerous advantages over current devices:
Summary of Benefits
- Allows reposition of device without losing eye contact of view screen
- Maintain absolute control of wire during device repositioning
- Increased back support during wire manipulation
- Reduction in wire retraction or advancement due to loss of wire control
- Shorter fluoro time
- Lower radiation exposure to physician and Cath Lab Staff
